The Hidden Risks of Improper Instrument Processing
We’re talking about risk analysis, the process we go through before deciding on a given action. Whether it’s diet soda or donuts we'll do a casual risk analysis in our head.
Non-Chemical Critical Water for Healthcare Reprocessing
As we prepare to attend AAMI next week, we see an important opportunity to strengthen shared understanding of how water treatment decisions influence safety, compliance, and sterilization outcomes in healthcare reprocessing. Case Medical, recognized as a U.S. EPA Safer Choice Partner of the Year, approaches chemical safety as an essential part of protecting both staff and patients.
Centralized Sterile Processing: Opportunity, Risk, and the Path Forward
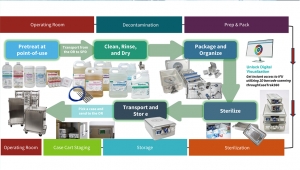
Reprocessing medical devices is a complex, multistep workflow that includes cleaning, inspection and assembly, functional testing, disinfection when required, packaging and labeling, sterilization, transport, and storage.
Rethinking Sterile Processing
Healthcare no longer operates in yesterday’s world. The Industrial Revolution has given way to the Digital Revolution, transforming how we diagnose, treat, and operate. Minimally invasive procedures now rely on sophisticated endoscopes and robotic instruments — devices that demand meticulous, step-by-step reprocessing and strict adherence to IFUs to ensure safety, performance, and longevity.
The KISS Principle for Instrument Processing
The last few blogs focused on the importance of standardization to decrease waste, remove unnecessary steps, improve turnover, and provide consistent workflow and increase efficiency. Currently, standardization and offsite processing are key buzz words for strategic plans to reduce handling and cut costs in SPD.
Standardization: The Missing Link in OR–SPD Communication
This week at the OR Leadership Summit, Case Medical connected with OR leaders to explore what it truly takes to achieve smoother operations, clearer communication, and reliable compliance. When communication breaks down between the operating room and sterile processing, the symptoms are easy to spot—late trays, incomplete sets, urgent phone calls, and last-minute workarounds.
Driving Better Outcomes Through Healthcare Standardization
Standardization can improve productivity, reduce costs, and create supply chain efficiency. Healthcare facilities are looking at the Total Cost of Ownership to reduce waste and have what is needed in stock, even during challenging times and supply chain shortages.
Penny Wise and Pound Foolish
When budgets are tight, people sometimes look for quick fixes to save on immediate costs. Unfortunately, they will end up being sorry if what looks like a less expensive product costs more in the long run.
Assessment Tools Foster Best Practices
Last month, Case Medical had another ISO Audit. We have them approximately two times a year, and it keeps us on our toes. As an ISO 13485 certified medical device manufacturer and an ISO 27001 certified software developer, a scheduled ISO audit and assessment ensures that we follow best practices...
The Healthcare System Poly Crisis and How Case Medical Can Help
If you think we are living in a time dominated by a climate crisis, a migration crisis, or a healthcare delivery crisis, you're mistaken; we are facing a poly crisis, the simultaneous occurrence of several catastrophic events that are nearly impossible to solve.